Design and created by Guideline Central in participation with the American Society of Clinical Oncology.

Metastatic Castration-Resistant Prostate Cancer
American Society of Clinical Oncology
Publication Date: January 20, 2026
Key Points
- Metastatic prostate cancer is a major cause of disease burden in patients, representing an estimated 313,780 new cases (the highest incidence in males, representing 30% of all new cases) and being responsible for 35,770 deaths (the second highest cause of mortality in males, representing 11% of all cancer-related deaths) in 2025.
- This guideline addresses questions on diagnostics, genomic testing, treatment, and specifically treatment options for small cell, neuroendocrine, and poorly differentiated prostate cancer.
Treatment
Principles of Practice
Principles of Practice Statement 1
Principles of Practice Statement 2
Principles of Practice Statement 3
Principles of Practice Statement 4
Principles of Practice Statement 5
Principles of Practice Statement 6
Principles of Practice Statement 7
Patients Previously Treated With ADT Alone in Castration Sensitive or Nonmetastatic Castration-Resistant Prostate Cancer (CRPC) Setting and Whose Disease Has Progressed to mCRPC
Recommendation 1.1.1
Recommendation 1.1.2
Recommendation 1.1.3
Recommendation 1.1.4
Recommendation 1.1.5
Recommendation 1.1.7
Figure 1. (Updated) Patients Previously Treated With ADT Alone

Patients Previously Treated With ADT and an ARPI and Whose Disease Has Progressed to mCRPC
Recommendation 1.2.1
Recommendation 1.2.2
Recommendation 1.2.2.1
Recommendation 1.2.3
Figure 2. (Updated) Patients Previously Treated With ADT and an ARPI

Patients Previously Treated With ADT and Docetaxel, and Whose Disease Has Progressed to Metastatic CRPC
Recommendation 1.3.1
Recommendation 1.3.2
Figure 3. (Updated) Patients Previously Treated with ADT and Docetaxel

Patients Previously Treated With ADT, ARPI and Docetaxel, and Whose Disease Has Progressed to Have Progressive Metastatic CRPC
Recommendation 1.4.1
Recommendation 1.4.2
Recommendation 1.4.3
Recommendation 1.4.4
Figure 4. (Updated) Patients Previously Treated with ADT, ARPI, and Docetaxel

b Independent of other biomarkers.
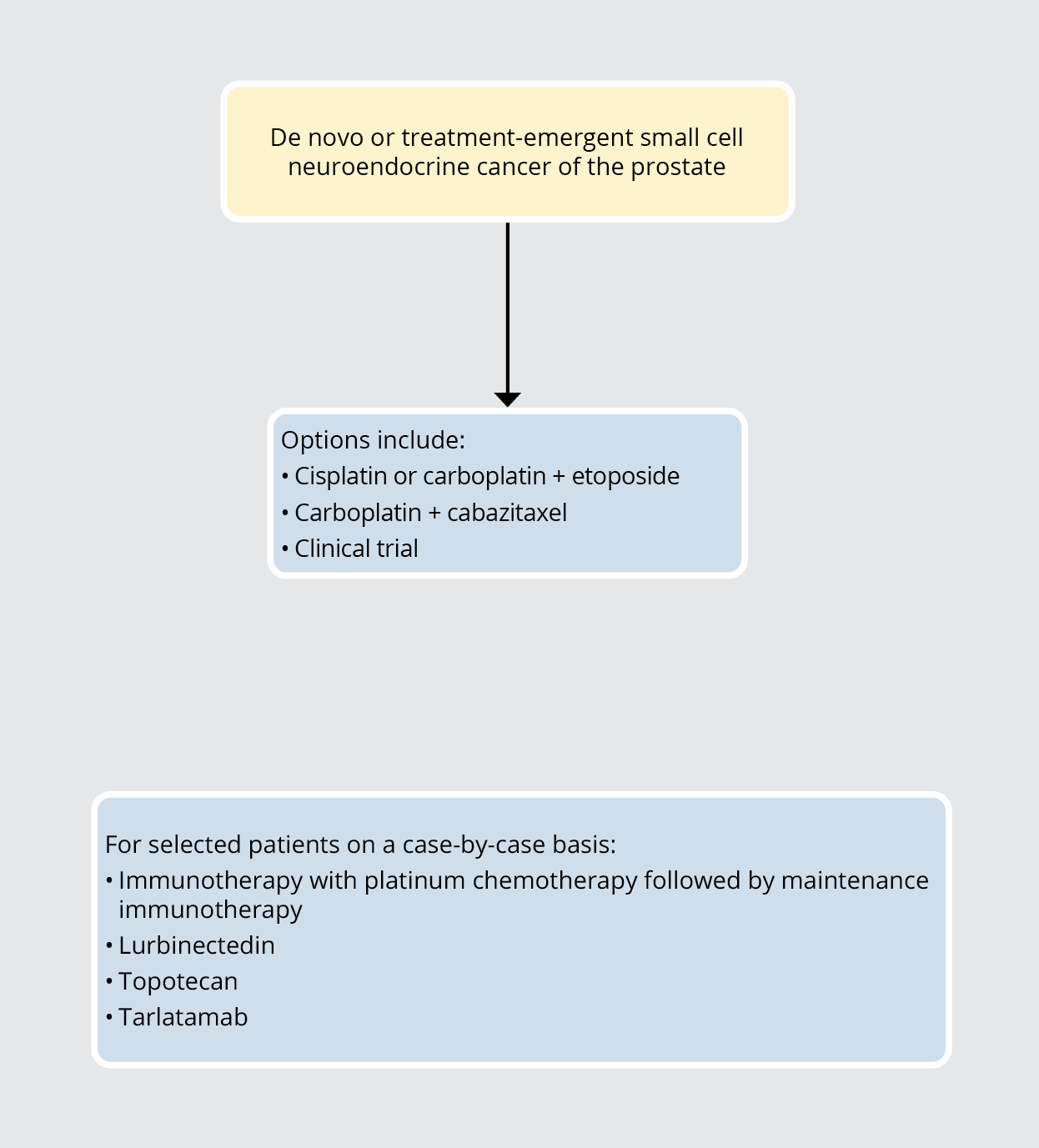
Treatment Options For De Novo or Treatment-Emergent Small Cell Neuroendocrine Carcinoma of the Prostate
Recommendation 2.1
Recommendation 2.2
Recommendation 2.3
Recommendation 2.3.1
Figure 5. Treatment Options for De Novo or Treatment-Emergent Small Cell Neuroendocrine Carcinoma of the Prostate
Assessing for Response While on Systemic Therapy for mCRPC
Recommendation 3.1
Recommended Scans for Response Assessment
Recommendation 3.2.1
Table 1. 10 Most Common Co-occurring Chronic Conditions Among Male Medicare Beneficiaries with Prostate Cancer (N = 1,016,617), 2011
| Beneficiaries less than 65 years (n = 26,243) | Beneficiaries 65 years and older (n = 990,374) | ||||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Prostate cancer prevalence | 0.9 | Prostate cancer prevalence | 8.8 | ||
| Top 10 co-morbidities | Top 10 co-morbidities | ||||
| Hypertension | 17,767 | 67.7 | Hypertension | 693,283 | 70 |
| Hyperlipidemia | 13,589 | 51.8 | Hyperlipidemia | 590,154 | 59.6 |
| Diabetes | 9,982 | 38 | Ischemic heart disease | 457,844 | 46.2 |
| Ischemic heart disease | 9,291 | 35.4 | Anemia | 337,957 | 34.1 |
| Anemia | 8,478 | 32.3 | Diabetes | 312,519 | 31.6 |
| Arthritis | 7,833 | 29.9 | Arthritis | 308,537 | 31.2 |
| Chronic kidney disease | 6,286 | 24 | Chronic kidney disease | 239,960 | 24.2 |
| Depression | 6,162 | 23.5 | Cataract | 235,241 | 23.8 |
| Chronic obstructive pulmonary disease (COPD) | 4,646 | 17.7 | Heart failure | 203,231 | 20.5 |
| Heart failure | 4,636 | 17.7 | COPD | 145,818 | 14.7 |
Notes:
Prepared by Information Products Group (IPG)/Office of Information Products and Data Analytics (OIPDA) on May 15, 2013.
Data Source: CMS administrative claims data, January 2011–December 2011, from the Chronic Condition Warehouse (CCW), ccwdata.org.
Population: Male Medicare beneficiaries enrolled in fee-for-service (FFS) coverage of both Parts A and B for the entire year. Beneficiaries who were enrolled at any point during the year in a Medicare Advantage (MA) plan were excluded as were beneficiaries who first became eligible for Medicare after January of the calendar year. Beneficiaries who died during the year were included up to their date of death if they meet the other inclusion criteria. Beneficiaries less than 65 years of age are primarily receiving Medicare due to a disability.
Chronic Condition Measures: For these tables, chronic conditions were identified through Medicare administrative claims. Medicare beneficiaries were considered to have a chronic condition if the CMS administrative data had a claim indicating that they were receiving a service or treatment for the specific condition. The data tables include information for beneficiaries with one of twenty-eight chronic conditions identified in the CCW. Detailed information on the identification of chronic conditions in the CCW is available at: http://www.ccwdata.org/chronic-conditions/index.htm.
The list of 28 conditions include:
Acquired hypothyroidism, Acute myocardial infarction, Alzheimer's Disease (including related Disorders or senile dementia), Anemia, Asthma, Atrial fibrillation, Autism, Benign prostatic hyperplasia, Breast cancer, Colon cancer, Endometrial cancer, Lung cancer, Prostate cancer, Cataract, Chronic kidney disease, COPD, Depression, Diabetes, Glaucoma, Heart failure, Hip/pelvic fracture, Hypertension, Hyperlipidemia, Ischemic heart disease, Osteoporosis, Arthritis (OA and RA), Schizophrenia (and other psychotic disorders) and Stroke.
Table 2. Costs
| Drug | Dosage | Form | Average Wholesale Price (AWP) | Price per |
| Abiraterone acetate* | 250 mg | Tablet | $1.60–97.21 | Each |
| Abiraterone acetate* | 500 mg | Tablet | $195.51–206.85 | Each |
| Abiraterone (micronized formulation) | 125 mg | Tablet | $104.57 | Each |
| Abiraterone acetate/niraparib | 50–500 mg, 100–500 mg | Tablet | $375.00 | Each |
| Apalutamide | 60 mg | Tablet | $149.13 | Each |
| Apalutamide | 240 mg | Tablet | $596.51 | Each |
| Darolutamide | 300 mg | Tablet | $135.74 | Each |
| Enzalutamide | 40 mg | Tablet, capsule | $143.32 | Each |
| Enzalutamide | 80 mg | Tablet | $286.65 | Each |
| Olaparib | 100 mg, 150 mg | Tablet | $168.54 | Each |
| Rucaparib | 200 mg, 250 mg, 300 mg | Tablet | $173.70 | Each |
| Talazoparib** | 0.1 mg, 0.25 mg, 0.35 mg, 0.5 mg | Capsule | $721.58 | Each |
| Sipuleucel-T | 50 million cells/250 mL | Suspension, intravenous | $300.49 | mL |
| 177Lu-PSMA (vipivotide tetraxetan) | 1000 MBq/mL (27 mCi/1 mL) | Solution, intravenous | $54,621.00 | mL |
| Radium 223 (223Ra-dichloride) | 1100 kBq/mL (30 microcurie/mL) | Solution, intravenous | $35,051.76 | mL |
| Docetaxel* | 10 mg/mL, 20 mg/mL | Solution, intravenous | $23.40–$365.15 | mL |
| Cabazitaxel | 60 mg/1.5 mL | Solution, intravenous | $11,317.23 | mL |
| Pembrolizumab | 100 mg/4 mL | Solution, intravenous | $1,700.61 | mL |
* Price range due to multiple manufacturers of generic.
** Would always have additional cost of enzalutamide.
Recommendation Rating Definitions
| Quality of Evidence | ||
|---|---|---|
| Term | Definitions | |
| H | - High | We are very confident that the true effect lies close to that of the estimate of the effect |
| M | - Moderate | We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
| L | - Low | Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect. |
| VL | - Very Low | We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect. |
| Ins | - Insufficient | Evidence is insufficient to discern the true magnitude and direction of the net effect. Further research may better inform the topic. Reliance on consensus opinion of experts may be reasonable to provide guidance on the topic until better evidence is available. |
| Strength of Recommendation | ||
|---|---|---|
| Term | Definitions | |
| S | - Strong | In recommendations for an intervention, the desirable effects of an intervention outweigh its undesirable effects. In recommendations against an intervention, the undesirable effects of an intervention outweigh its desirable effects. All or almost all informed people would make the recommended choice for or against an intervention. |
| C W | - Conditional - Weak | In recommendations for an intervention, the desirable effects probably outweigh the undesirable effects, but appreciable uncertainty exists. In recommendations against an intervention, the undesirable effects probably outweigh the desirable effects, but appreciable uncertainty exists. Most informed people would choose the recommended course of action, but a substantial number would not. |
| GPS | Good Practice Statement | Good practice statements represent the consensus of the Expert Panel and are used when high quality indirect evidence is available, but it would not be a good use of the Expert Panel’s resources to conduct a formal systematic review |
| NR | - Not Rated | |
Brozek JL, Akl EA, Compalati E, et al: Grading quality of evidence and strength of recommendations in clinical practice guidelines part 3 of 3. The GRADE approach to developing recommendations. Allergy 66:588-95, 2011
Source Citation
Taplin M, Riaz IB, Rumble RB, et al. Systemic Therapy in Men with Metastatic Castration Resistant Prostate Cancer: ASCO Living Guideline Update, Version 2025.1. J Clin Oncol. 2026 Jan 20. doi: 10.1200/JCO-25-02693
Garje R, Riaz IB, Naqvi SA, et al. Systemic Therapy in Men with Metastatic Castration Resistant Prostate Cancer: ASCO Guideline Update. J Clin Oncol. 2025 May 2. doi: 10.1200/JCO-25-00007
Disclaimer
This pocket guide is derived from recommendations in the American Society of Clinical Oncology Guideline. This resource is a practice tool based on ASCO® practice guidelines and is not intended to substitute for the independent professional judgment of the treating physician. Practice guidelines do not account for individual variation among patients. This pocket guide does not purport to suggest any particular course of medical treatment. Use of the practice guidelines and this resource are voluntary. The practice guidelines and additional information are available at www.asco.org/ genitourinary-cancer-guidelines. Copyright © 2025 by American Society of Clinical Oncology. All rights reserved.
Guideline Central and select third party use “cookies” on this website to enhance the user experience.
This technology helps us gather statistical and analytical information to optimize the relevant content for you.
The user also has the option to opt-out which may have an effect on the browsing experience.